Since the U.S. Food and Drug Administration approved the abortion pill Mifeprex in 2000, this lethal drug has been used to kill nearly 4 million American children and has resulted in at least 24 maternal deaths. Now, under the guise of mitigating exposure to COVID-19, the abortion lobby has successfully blocked the FDA from enforcing its minimal requirements designed to ensure that this drug does not also kill or injure the women who ingest it.
The abortion industry wants these FDA requirements, called REMS (Risk Evaluation and Mitigation Strategy) permanently dropped. An Obama-appointed judge’s decision on July 13 to enjoin the FDA from enforcing REMS was a strategic step toward making the abortion pill available over the counter or online.
Unlike the “morning-after pill,” Mifeprex was designed with the singular purpose of killing a developing child after pregnancy has been confirmed. The abortion pill is used to cause a “chemical” or “medication” abortion and is taken under a regimen consisting of two FDA-approved drugs: mifepristone (Mifeprex) and misoprostol. Until recently, the use of misoprostol for abortion was considered “off-label.”
How the Pill Works
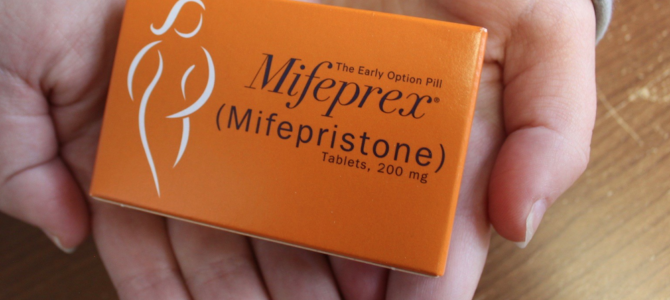
According to protocol, the pregnant woman first takes one 200-milligram tablet of Mifeprex orally. Mifeprex kills the developing child by blocking the critical pregnancy hormone progesterone. As the uterine lining degenerates from lack of progesterone, the preborn child dies from lack of oxygen and other essential nutrients.
If a woman changes her mind about going through with the abortion after taking Mifeprex, but prior to taking the misoprostol, she can take progesterone to counteract the effects of Mifeprex. Time is of the essence in these cases to save the baby’s life. A recent study of babies rescued by this method found no increase in birth defects.
If the woman seeks to complete the abortion-pill regimen, she will take four 200-milligram tablets of misoprostol buccally (placed in her cheek), 24 to 48 hours after taking Mifeprex. Misoprostol is a prostaglandin designed to treat stomach ulcers.
It is used to cause uterine cramping and contractions, artificially inducing labor and heavy bleeding to expel the child from her body. Misoprostol thus induces symptoms normally associated with miscarriage. Any experienced OB/GYN can attest to the life-threatening bleeding and other complications associated with miscarriage.
The FDA permits the abortion pill’s use during the first 10 weeks of pregnancy, extended from seven weeks by the Obama administration in 2016. A baby grows rapidly in the first 10 weeks. The human heartbeat can first be detected about three weeks after fertilization. In the following weeks, eyes develop and brain activity can be recorded. By 10 weeks of gestation, the embryo is an inch and a quarter long, all vital organs are in place, and teeth begin to harden and connect to the jawbone.
This Drug Is Deadly
The normal side-effects of this abortion pill range from serious to deadly. Heavy bleeding can turn into hemorrhaging, which can be fatal. A woman can also retain parts of her baby or placenta after the abortion process, causing serious or fatal infections.
Holly Patterson had just turned 18 when she took Mifeprex. Four days into her abortion process, Holly called the abortion facility that administered the drugs, complaining of severe pain. She was told to take a painkiller and go to the emergency department if her symptoms worsened — and they did.
At the emergency department, doctors gave her narcotic pain medication and sent her home, where her pain continued. Seven days into the abortion process, Holly went back to the hospital, where she died of Clostridium sordellii toxic shock syndrome.
This drug is so dangerous that the REMS only permits it to be dispensed in “healthcare settings” such as “clinics, medical offices, and hospitals.” Prescribers must be approved after signing a Prescriber Agreement guaranteeing they are capable of accurately dating the pregnancy and diagnosing dangerous ectopic pregnancies. Prescribers must further agree to provide or arrange for “surgical intervention” in case of failure or complications.
The REMS also requires that each woman sign a Patient Agreement and receive extensive written disclosures about the drug regimen’s side-effects. The woman must attest: “If I need a surgical procedure because the medicines did not end my pregnancy or to stop heavy bleeding, my healthcare provider has told me whether they will do the procedure or refer me to another healthcare provider who will.”
Pull the Pill from Market
Given the quantity and nature of the adverse events reported to the FDA since 2000, it is unconscionable that the FDA has not pulled Mifeprex off the market. The abortion lobby now seeks to expand access to this lethal drug by using the federal courts, and convincing judges with no medical training to circumvent the FDA’s oversight.
Prescribing the abortion pill entirely by telemedicine poses additional health risks to women. First, life-threatening ectopic pregnancies will surely be missed. Second, there will be no way to confirm a woman’s pregnancy dates unless a pelvic ultrasound is ordered, which defeats the purpose of allowing these drugs to be prescribed by telemedicine in the first place.
Women are often wrong about their dates, and without the requirement for an in-person exam or ultrasound, women may inadvertently — or purposefully — request this regimen beyond 10 weeks of pregnancy. Taking this regimen beyond 10 weeks can result in disastrous consequences for women and their babies.
The abortion pill is explicitly designed and prescribed to destroy preborn children and thus will never be “safe” for women or the preborn children it is designed to kill. In the abortion industry’s ruthless pursuit of profit, it treats the lives of millions of unborn babies and dozens of women as collateral damage. The abortion industry will not take its foot off the accelerator voluntarily.
We insist the FDA exercise its authority to classify the abortion pill as an “imminent hazard to the public health” that poses a “significant threat of danger” and remove this lethal drug from the U.S. market.