When it comes to distribution of the abortion drug mifepristone, the U.S. Food and Drug Administration (FDA) has been asleep at the switch. Despite comprehensive data showing higher rates of complications and emergency room visits from chemical abortion, the abortion pill has been feted by the corporate media, promoted as “safe” by President Joe Biden, and subjected to increasingly lax oversight by the FDA.
All of this makes a little-heralded recent story in Politico even more interesting. An anonymous FDA source warns against the intensifying abortion industry campaign to give abortion pills to women and girls who are not pregnant. Politico quotes the unnamed FDA spokesman expressing concern that this approach poses new health risks to women: “Mifepristone is not approved for advance provision of a medical abortion.”
The anonymous source goes on to list several safety concerns including the inability of doctors to screen for ectopic pregnancy or properly date a pregnancy (abortion pill risks increase exponentially with each week of pregnancy). To that list, we add the very real risk of sex traffickers or abusive partners using careless distribution of abortion pills to commit crimes of coercion. The Charlotte Lozier Institute has been sounding the alarm on these risks for years, though often falling on deaf federal ears.
There is little sign that the abortion industry cares about these concerns, as even the FDA has given mifepristone glancing affirmation in recent years. In fact, the FDA has been in more or less full retreat from its oversight of mifepristone.
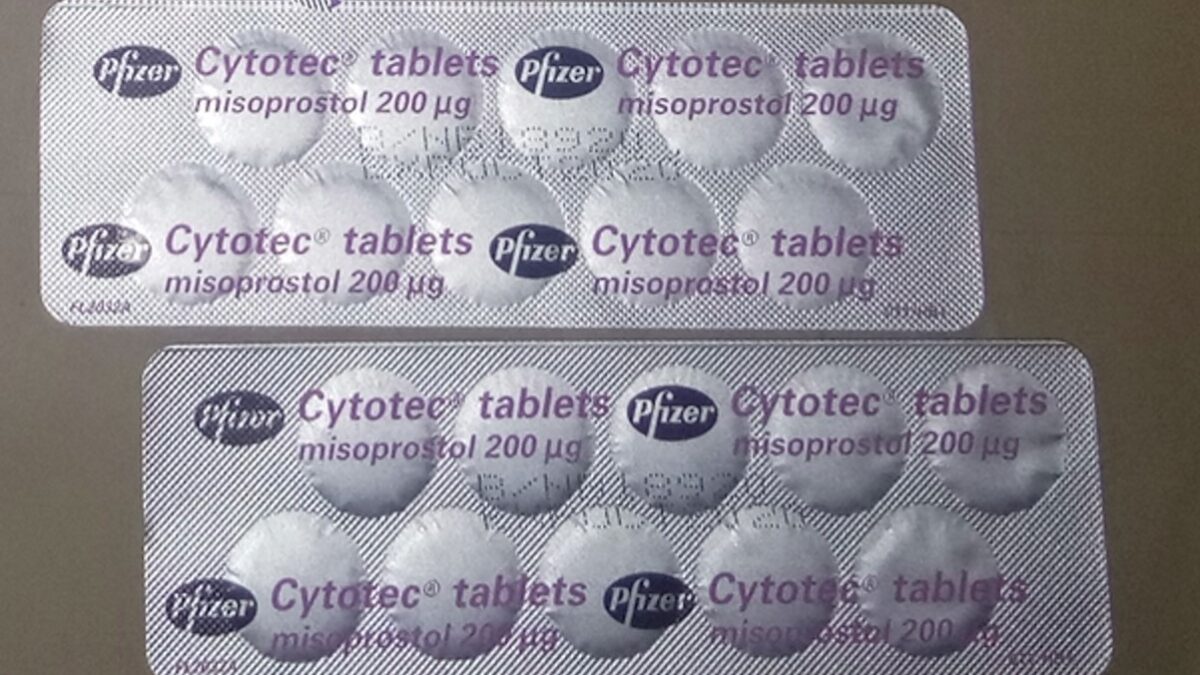
The abortion drug regimen of mifepristone (to kill the baby) and misoprostol (to expel the dead baby) was first approved in the waning days of the Clinton administration under a regulation designed to reduce the risks associated with hazardous drugs. Known as ETASU, Elements To Assure Safe Use, the regulation was meant to ensure women obtain abortion drugs from a certified physician capable of monitoring and treating complications. For abortion pills, those complications range from death to hemorrhage to infection, requiring visits to the emergency room and, in many cases, follow-up surgery.
FDA Loosened Requirements
The initial FDA rules also required reporting and tracking these complications, which proved to be wise. Mifepristone’s original mode of administration was found to be associated with lethal infections. Other complications proved more frequent than the rate associated with surgical abortion procedures.
How did the FDA react? It changed the recommended procedure for administration, but oddly enough in March 2016, the FDA removed the requirement that only a physician could prescribe the drug and dropped any prescriber requirement for reporting non-fatal complications, the exact opposite of what one would expect the FDA would do to assure safe use. The goal was to widen distribution of the abortion pill, while learning less from its manufacturers about the dangerous complications the new policy might cause.
Use of mifepristone-misoprostol accelerated after 2016, to the point where today it is the method deployed in the majority of reported abortions. Taking advantage of the declared Covid emergency, the pro-abortion American College of Obstetricians and Gynecologists brought suit in a friendly federal court and won suspension of the in-person dispensing requirement, allowing the drugs to be distributed via telehealth and the mail. The ruling was reversed by the Supreme Court in late 2020, but not for long.
Then the Biden administration, well prior to the Supreme Court decision in the Dobbs v. Jackson Women’s Health Organization case, paved the way for abortion pills by mail, a prelude perhaps to selling the drugs over the counter. In April 2021, the FDA announced it was suspending the in-person dispensing requirement under the guise of the Covid “emergency.” In December 2021, the FDA decided to permanently allow abortion by mail.
Profoundly Anti-Woman Implications
The implications of these policies are profoundly anti-woman and anti-family. Research based on 17 years of Medicaid claims data analyzed by the Charlotte Lozier Institute shows that chemical abortions are a growing public health threat, with the rate of abortion pill-related emergency room visits spiking more than 500 percent from 2002 to 2015. The need to utilize claims data reflects the lack of FDA reporting requirements, exceptionally weak state abortion reporting laws, and only voluntary federal reporting.
The advent of “no-test” administration of mifepristone only compounds the risks to women and girls. Without physical examination, ultrasound, labs, or a physician-patient relationship, women can obtain abortion pills through the mail or telehealth and commence an abortion, absent the confirmation of an intrauterine pregnancy, correct gestational age, and determination of potential risk factors for future pregnancy complications.
Imagine being a parent and discovering that your 15-year-old daughter has somehow obtained mifepristone and misoprostol via her school, a friend, or the internet. Imagine she is one of the women who endure up to 16 days of hemorrhaging from the drug, or that she is further along than the 10 weeks of pregnancy for which the drug is authorized. Imagine she delivers an extremely premature baby — which by 10 or 11 weeks of gestation will have fingers and toes and more than 4,000 distinct body parts — in the family bathroom, alone, frightened, and weighted with an unerasable memory.
This is increasingly the state of abortion in America. The abortion industry has steadily decreased expert medical care for pregnant women, ignored all standards of prudence, isolated the young without family or counseling support — and, adding insult to injury — devised distribution schemes for abortion pills that will both foster and rely on the absence of public health oversight. The FDA is right to finally be alarmed, but the hour is late.